Carbon
Carbon is a very abundant element. It is so important to life that we dedicated a branch of chemistry which specifically deals with it. This is called organic chemistry. Carbon is found in all organic molecules, and almost all compounds containing carbon are considered organic. Organic molecules make up almost everything in our bodies: proteins, fats, nucleic acids, etc.
But what makes carbon so special? Well, carbon has 6 electrons in total, 2 of them are in the first electron shell, so that leaves 4 electrons in its valence shell, so it can form 4 covalent bonds. This is a lot compared to hydrogen's 1, oxygen's 2, and nitrogen's 3. The fact that carbon can form bonds with so many partners makes it the backbone for large molecules. Other than carbon, silicon can also form 4 bonds, but it is much less common on Earth, this is why life as we know it is based on carbon.
But what makes carbon so special? Well, carbon has 6 electrons in total, 2 of them are in the first electron shell, so that leaves 4 electrons in its valence shell, so it can form 4 covalent bonds. This is a lot compared to hydrogen's 1, oxygen's 2, and nitrogen's 3. The fact that carbon can form bonds with so many partners makes it the backbone for large molecules. Other than carbon, silicon can also form 4 bonds, but it is much less common on Earth, this is why life as we know it is based on carbon.
Isomers
Isomers are molecules that have the exact same atoms but arranged differently. Even though they are made of the same elements, most isomers differ greatly in structure. The locations of functional groups around the carbon chain causes this. There are 3 common types of isomers:
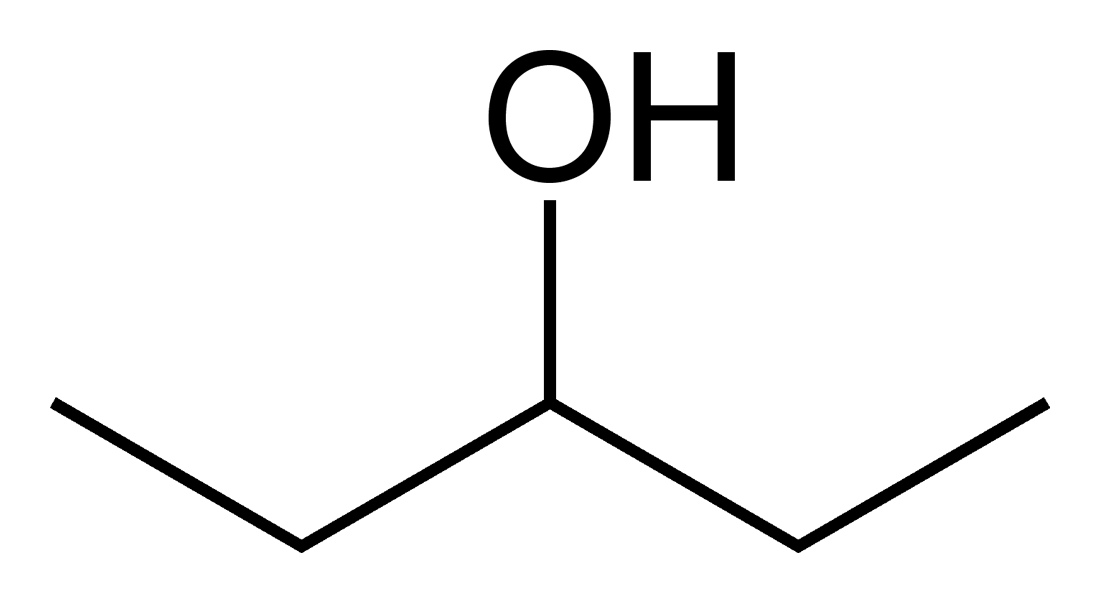
Structural isomers: They differ in where bonds are formed and/or the location of double bonds.
Structural isomers: They differ in where bonds are formed and/or the location of double bonds.
These above pentanol's all have the same number and type of atoms. However, the location of the OH group varies.
Geometric Isomers: They differ in arrangement of branches about a double bond. If the branches are on the same side, then it's called a cis isomer. If the branches are on the opposite sides, then it's called a trans isomer.
Enantiomers: They differ in arrangement around a carbon with asymmetric atoms around it. This results in molecules that are mirror images of each other.
Despite having almost identical structure to each other, isomers vary greatly in function. Even the difference in one atom's placement can dramatically alter the function.
Functional Groups
Functional groups are the molecular attachments to the carbon backbone. Different functional groups have vastly different properties. This is another reason why diversity exists among biomolecules.
Hydroxyl: A hydrogen atom is bonded to an oxygen atom, which is then attached to the carbon skeleton.
Carbonyl: An oxygen atom is double bonded to a carbon atom in the carbon chain. When this happens at the end of the carbon chain it is called an aldehyde. When this happens anywhere else in the carbon chain it is called a keytone.
Carboxyl: An oxygen atom is double bonded to a carbon atom (just like in carbonyl). But the carbon atom is also bonded to a hydroxyl group. We combine the names of carbonyl and hydroxyl to form carboxyl. They can donate hydrogen ions, thus they act as an acid.
Amino: A nitrogen atom is bonded to two hydrogen atoms. The nitrogen is also connected to the carbon chain. Amines accept hydrogen ions, so they function as a base.
Sulfhydryl: A sulphur atom is bonded to a hydrogen atom and to the carbon chain.
Phosphate: A phosphorus atom is bonded to four oxygen atoms, one of which is bonded to the carbon chain.
Hydroxyl: A hydrogen atom is bonded to an oxygen atom, which is then attached to the carbon skeleton.
Carbonyl: An oxygen atom is double bonded to a carbon atom in the carbon chain. When this happens at the end of the carbon chain it is called an aldehyde. When this happens anywhere else in the carbon chain it is called a keytone.
Carboxyl: An oxygen atom is double bonded to a carbon atom (just like in carbonyl). But the carbon atom is also bonded to a hydroxyl group. We combine the names of carbonyl and hydroxyl to form carboxyl. They can donate hydrogen ions, thus they act as an acid.
Amino: A nitrogen atom is bonded to two hydrogen atoms. The nitrogen is also connected to the carbon chain. Amines accept hydrogen ions, so they function as a base.
Sulfhydryl: A sulphur atom is bonded to a hydrogen atom and to the carbon chain.
Phosphate: A phosphorus atom is bonded to four oxygen atoms, one of which is bonded to the carbon chain.
Notice how all of these functional groups attach onto the carbon chain. This further illustrates carbon's importance in organic chemistry.